Most engineers inherit plating specifications from legacy designs or rely on what is commonly used in their industry for similar applications. But here's the challenge: Do those standards actually validate what your part needs to do in the field?
Why Tri-M3TM Tri-Alloy Plating Is Enabling Next-Generation Technology Performance (7 Use Cases)
Engineers developing next-generation RF connectors, telecommunications infrastructure, and high-frequency components face a persistent tradeoff: silver delivers superior electrical conductivity but tarnishes rapidly and drives up costs, while nickel offers durability and corrosion resistance but introduces magnetic interference that degrades signal quality. This compromise has constrained design decisions for decades, forcing manufacturers to choose between suboptimal performance and elevated material costs.
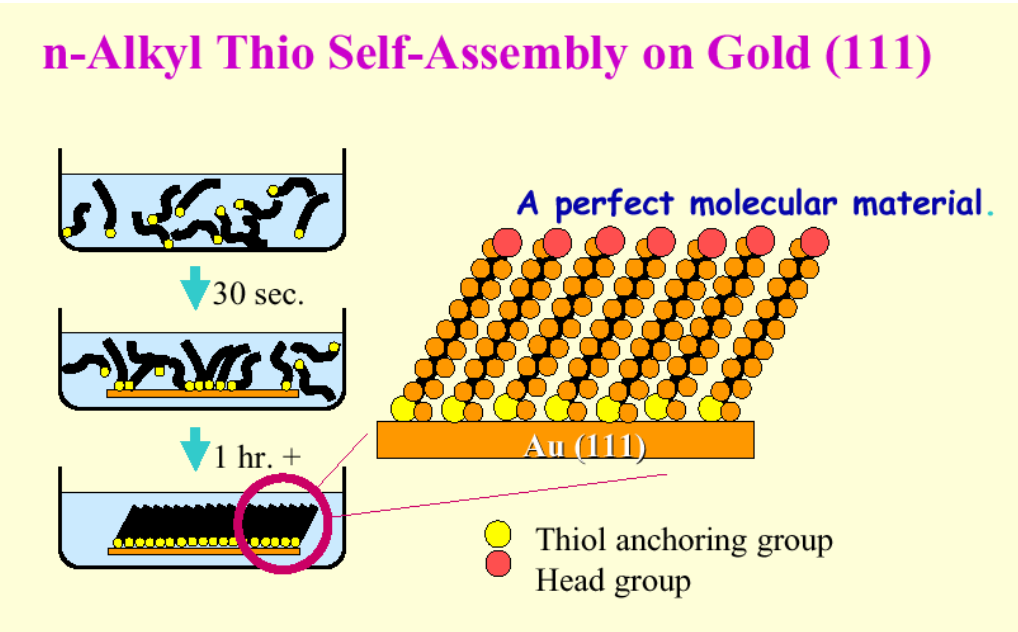
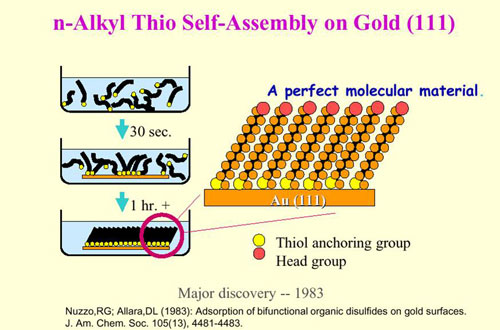
As manufacturing industries push the boundaries of miniaturization, performance, and cost efficiency, traditional surface finishing approaches face mounting limitations. Components in aerospace, telecommunications, medical devices, and advanced electronics require surfaces that deliver exceptional performance characteristics while meeting increasingly stringent dimensional tolerances. Self-assembled monolayers (SAMs) represent a breakthrough approach to surface engineering that addresses these challenges through molecular-level precision.
You've specified the perfect plating material. Your part geometry is optimized. The service provider follows all the right standards. Yet when the parts arrive, you discover adhesion failures, contamination issues, or inconsistent coverage that compromises performance and forces expensive rework.
When it comes to high-reliability components, plating isn’t just the finishing touch—it’s a critical performance factor. But for parts with complex geometries, achieving consistent, functional, and spec-compliant plating can be far from simple.
Rework and scrap pose critical challenges to manufacturers, particularly in industries requiring precision and reliability. Defective or out-of-spec components lead to increased costs, production delays, and strained supplier relationships. For safety-critical components in highly regulated industries like aerospace and automotive, these issues are especially damaging, creating bottlenecks that disrupt the supply chain and delay deliveries to OEMs.
A wide range of devices with numerous applications use precious metal plating. This process, however, can often be expensive and the precious metal is subjected to wear and tear that can eventually cause failures. Electro-Spec’s innovative methods for plating include a technological advancement known as NanoSHIELD-AU. The NanoSHIELD-AU process allows, for example a gold plated component to have a much thinner deposit without a reduction in functionality.
What is NanoSHIELD-AU?
SAM is an acronym for Self-Assembled Molecules. The NanoSHIELD-AU process employs bi-Functional or multi-functional molecules that offer two or more termination groups with different functionality. Essentially, the NanoSHIELD-AU process uses molecules that offer two levels of protection. The molecules used in the NanoSHIELD-AU method can be attached to metal alloys as well as ceramic, glass, plastics, and more. Right now, NanoSHIELD technology is only being used as a post-plate surface treatment for gold and tri-alloy (Tri-M3).
Products Finishing magazine names Electro-Spec to the ‘Top Shops” list
CINCINNATI, Ohio – Electro-Spec, Inc. has been named one of the best finishing shops in the U.S., according to an industry benchmarking survey conducted by Products Finishing magazine, a trade publication which has covered the industry since 1938.
Plating protects metal surfaces, increasing their hardness, tensile strength and applies an aesthetic quality. When seeking appropriate plating for a specific project, you’ll find that there are a number of nickel plating types and options to consider.
So how does one determine exactly which plating is best for your particular situation? The first item to consider is the application.
Generally speaking, if a conductive surface doesn’t require a high corrosion or wear resistance but needs a bright sheen or low-stress layer, then electrolytic plating will be the most efficient process.
Silver plating started as a common way to provide cheaper versions of household items, which were originally made of silver.
At its beginning in the 18th century, this included cutlery, platters, plates and candlesticks among other items. In the 19th century, electroplating arose as a rapid method of finishing mass-produced items. Although the 20th and 21st centuries have seen declines in the use of silver for household wares, this reduction was followed by a rise in the use of silver plating for the electronics industry.